|
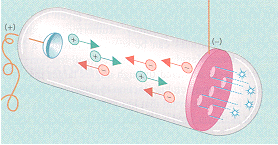
When the cathode of a cathode-ray tube was perforated, Goldstein observed rays he called "canal rays," which passed through the holes, or channels, in the cathode to strike the glass walls of the tube at the end near the cathode. Since these canal rays travel in the opposite direction from the cathode rays, they must carry the opposite charge. |