Grignard Reagents
So far, we have built a small repertoire of reactions that can
be used to convert one functional group to another. We have
briefly discussed converting alkenes to alkanes; alkanes to alkyl
halides; alkyl halides to alcohols; alcohols to ethers,
aldehydes, or ketones; and aldehydes to carboxylic acids. We have
also shown how carboxylic acids can be converted into esters and
amides. We have yet to encounter a reaction, however, that
addresses a basic question: How do we make C![]() C bonds? One
answer resulted from the work that Francois Auguste Victor
Grignard started as part of his Ph.D. research at the turn of the
century.
C bonds? One
answer resulted from the work that Francois Auguste Victor
Grignard started as part of his Ph.D. research at the turn of the
century.
Grignard noted that alkyl halides react with magnesium metal in diethyl ether (Et2O) to form compounds that contain a metal-carbon bond. Methyl bromide, for example, forms methylmagnesium bromide.
| Et2O | ||
| CH3Br + Mg | CH3MgBr |
Because carbon is considerably more electronegative than magnesium, the metal-carbon bond in this compound has a significant amount of ionic character. Grignard reagents such as CH3MgBr are best thought of as hybrids of ionic and covalent Lewis structures.
![]()
Grignard reagents are our first source of carbanions (literally, "anions of carbon"). The Lewis structure of the CH3- ion suggests that carbanions can be Lewis bases, or electron-pair donors.
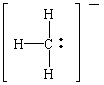
Grignard reagents such as methylmagnesium bromide are therefore sources of a nucleophile that can attack the + end of the C=O double bond in aldehydes and ketones.
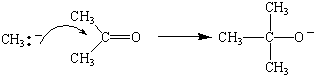
If we treat the product of this reaction with water, we get an tertiary alcohol.

If we wanted to make a secondary alcohol, we could add the Grignard reagent to an aldehyde, instead of a ketone.
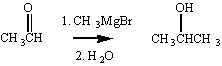
By reacting a Grignard reagent with formaldehyde we can add a single carbon atom to form a primary alcohol.
This alcohol can then be oxidized to the corresponding aldehyde.
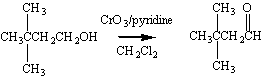
The Grignard reagent therefore provides us with a way of performing the following overall transformation.
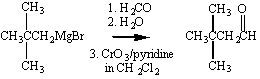
A single carbon atom can also be added if the Grignard reagent is allowed to react with CO2 to form a carboxylic acid.
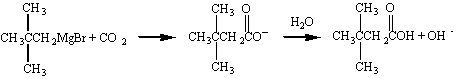
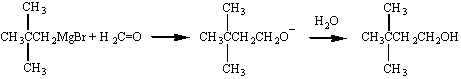
Perhaps the most important aspect of the chemistry of Grignard reagents is the ease with which this reaction allows us to couple alkyl chains. Isopropylmagnesium bromide, for example, can be used to graft an isopropyl group onto the hydrocarbon chain of an appropriate ketone, as shown in the figure below.
