Practice Problem 1
Elemental phosphorus is often stored under water because it doesn't dissolve in water. Elemental phosphorus is very soluble in carbon disulfide, however. Explain why P4 is soluble in CS2 but not in water.
Answer
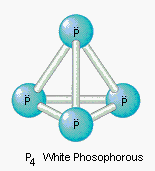
The structure of the P4 molecule is shown below.

This molecule is a perfect example of a nonpolar solute. It is therefore more likely to be soluble in nonpolar solvents than in polar solvents such as water.
The Lewis structure of CS2 suggests that this molecule is linear.
![]()
Thus, even it there is some separation of charge in the C=S double bond, the molecule would have no net dipole moment, because of its symmetry. The electronegativities of carbon (EN = 2.55) and sulfur (EN = 2.58), however, suggest that the C=S double bonds are almost perfectly covalent. CS2 is therefore a nonpolar solvent, which should readily dissolve P4.