- In the first beaker half-filled with 0.1M CuSO4 solution, zinc metal is added.
- In the second beaker half-filled with 0.1M ZnSO4 solution, copper metal is added.
- Iron metal is immerse in the 0.1M CuSO4 solution.
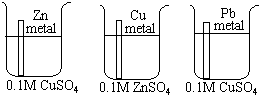
Chemical Concept Demonstrated: Oxidation/reduction reactions between metals and metal ions
Demonstration:
|
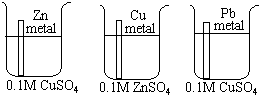 |
Observations:
The zinc metal is coated with dark-black elemental copper. The
copper metal, however, has no reaction with the ZnSO4 solution.
The iron metal is coated with red-bronze metallic copper.
Explanations (including important chemical equations):
| Zn (s) | + | CuSO4 (aq) | ----> | Cu (s) | + | ZnSO4 (aq) |
| stronger reducing agent | stronger oxidizing agent | weaker reducing agent | weaker oxidizing agent |
Cu (s) + ZnSO4 (aq) ---> Zn (s) + CuSO4 (aq)
Fe (s) + CuSO4 (aq) ---> Cu (s) + FeSO4 (aq)
Oxidation occurs when the oxidation number of an atom becomes larger. Reduction occurs when the oxidation number of an atom becomes smaller. Oxidizing agents gain electrons while reducing agents lose electrons. Generally, when metals are fully reduced, they form elemental metal and "plate" the site where they were reduced.
In the first reaction, the copper ion is able to oxidize the zinc metal. However, in the second reaction, the zinc ion is not able to oxidize the copper metal. Zinc is a better reducing agent than copper. Strong reducing agents have weak conjugate oxidizing agents. Zn2+ is a weak conjugate oxidizing agent compared to Cu2+. Conversely, strong oxidizing agents have weak conjugate reducing agents. The second reaction did not occur because the reactants were the weaker reducing and oxidizing agents. In the third reaction, iron is a better reducing agent than copper, however iron is not as good as zinc.