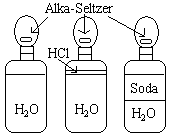
Three balloons are attached to three bottles as shown in the picture.
|
 |
Chemical Concepts Demonstrated: Chemistry of HCO3- ion, acid-base reactions, LeChatelier's Principle
Demonstration:
Three balloons are attached to three bottles as shown in the picture.
|
 |
Drop the alka-seltzer into the bottles.
Observations:
The more acidic the solution, the larger the amount of CO2 gas is produced. These amounts can easily be measured by comparing the resulting size of the balloons. The plain water produces the least amount of gas, the water and hydrochloric acid produces a moderate amount, and the water and soft drink produce the most gas.
Explanation (including important chemical equations):
HCO3- (aq) + H+ (aq) <=> H2CO3 (aq) <=> H2O (l) + CO2 (g)
HCl is a stronger acid than water. Larger amounts of acid will increase the acidity of the solution and push the reaction towards the production of products (i.e. more acidity yields more CO2 gas).