- The top layer is made up of HCl dissolved in toluene.
- The middle layer is universal indicator in water.
- The bottom layer is NH3 dissolved in chloroform (CHCl3).
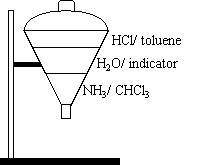
Chemical Concept Demonstrated: Diffusion of liquids
Demonstration:
100 mL of three solutions are poured into a large filter funnel, being careful not to
mix the layers.
|
 |
Observations:
The top HCl layer diffuses into the middle layer and changes color
indicating an acid. The bottom NH3 layer is also diffusing into the
middle layer and changes color indicating a base. Eventually once the HCl and the NH3
begin to mix, the indicator turns the color of a neutral solution.
Explanation:
Liquids diffuse from solutions of higher concentration to solutions of lower concentration. The indicator in the water showed how the HCl diffused downward into the water, but the NH3 diffused upward. Both were going toward the solution with the lowest concentration. Then, they began to mix with each other in the same manner.