One student takes the beaker, fills it with water, and transfers it to the opposite aquarium on the left.
The other student does this, transferring the water to the right.
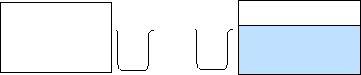
Chemical Concept Demonstrated: Dynamic equilibrium
Demonstration:
| One aquarium is 4/5 filled with water, the other is empty. One student takes the beaker, fills it with water, and transfers it to the opposite aquarium on the left. The other student does this, transferring the water to the right. |
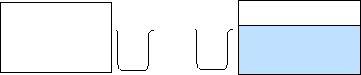 |
Observations:
The amount of "reactant" in one aquarium decrease with
time, while the amount of "product" in the other aquarium increases. Soon there
is no change in the water level between the two aquariums.
Explanation:
When the students start with the identical beakers, equilibrium is reached as soon as the level of water in the "product" aquarium reaches a point where the student obtains a full beaker of water each time the student dips into the aquarium. Exactly the same amount of water is transferred from left to right as from right to left. Regardless of the combination of beakers used in this demonstration, an equilibrium will be reached. The water level in each aquarium at equilibrium, however, depends of the size of the beakers used.