
Chemical Concept Demonstrated: How the concentration of components of a system of coupled equilibria changes as the conditions of the system are changed
Demonstration:
A small quantity of Fe3+solution is added to 8 crystallizing dishes.
|
Observations:
 |
Explanation (including important chemical equations):
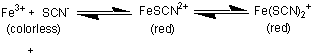 |
Dish Group 1, 4, 7: The Cit3- ion added to dishes 4, and 7 forms a light yellow FeCit complex. The yellow FeCit complex in dish 7 disappears after the addition of HClO4 due to the formation of H3Cit, H2Cit -, and HCit2-. They do not form complexes with Fe3+.
Dish Group 2, 5, 8: The KSCN added to dishes 2, 5, and 8 forms the blood red complex Fe(SCN)2+/ Fe(SCN)2+ complexes. Cit3- are added to dishes 5 and 8 to form a yellow FeCit complex in the place of the red Fe(SCN)2+/ Fe(SCN)2+ complexes. HClO4 is added to dish 8. H3Cit, H2Cit -, and HCit2- are formed, sequestering the Cit3- ion, and the solution turns blood red as the Fe(SCN)2+/ Fe(SCN)2+ complexes are formed.
Dish Group 1, 2, 3: The KSCN added to dish 3 also forms the blood red Fe(SCN)2+/ Fe(SCN)2+ complexes. HClO4 is then added to dish 3. HSCN is a strong acid. The SCN - is too weak a base for the formation of HSCN to interfere with the Fe(SCN)2+/ Fe(SCN)2+ complexes, so there is no apparent change in the color of the solution.
Dish Group 1, 2, 6: KSCN is also added to dish 6 to form the blood red Fe(SCN)2+/ Fe(SCN)2+ complexes. Ascorbic acid is then added and reduces the Fe3+ ion to Fe2+, which does not form complexes with SCN -.