- Aqueous ammonia is added dropwise into the first dish.
- Excess ammonia is added into the first dish.
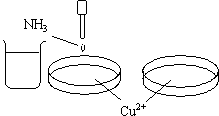
Chemical Concept Demonstrated: Complex-formation equilibrium
Demonstration:
Each of the dishes contain Cu 2+ ion solution.
|
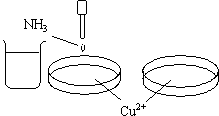 |
Observations:
A precipitate forms as the ammonia is added dropwise into the dish. Additional ammonia causes the precipitate to dissolve and a dark-blue solution is formed.
Explanations (including important chemical equations):
A light-blue colored precipitate of Cu(OH)2 is formed when aqueous ammonia is added to Cu 2+ ion solution.
Cu 2+ (aq) + 2 (OH)- (aq) <=> Cu(OH)2 (s) Ksp = 2.2 x 10-20
In the presence of excess NH3, this equilibrium shifts to the left as the free Cu 2+ ion is sequestered from the solution as the Cu(NH3)4 2+ complex ion.
Cu 2+ (aq) + 4 NH3 (aq) <=> Cu(NH3)4 2+ (aq) Ksp = 2.1 x 1013
The intensity of the absorption of light by the Cu(NH3)4 2+ complex appears to be orders of magnitude greater than the corresponding Cu(H2O)6 2+ complex. The molar absorbances of these complexes, however, differ, roughly, only by a factor of four. The difference between the intensities of the colors of these complexes can be explained by noting that the maximum wavelength for the Cu(H2O)6 2+ complex is about 800 nm, whereas the maximum wavelength for the Cu(NH3)4 2+ complex occurs at about 600 nm. Since a larger portion of the absorption in the Cu(NH3)4 2+ complex is in the visible portion of the spectrum, the color appears considerably more intense.