- Indigo carmine is added to a 1:1 mixture of ethanol and water.
- KOH is dissolved in water, dextrose (glucose) is added to this solution, along with the indicator solution.
Chemical Concept Demonstrated: Reversible oxidation-reduction reactions
Demonstration:
|
 |
Observations:
When the indicator is initially added to the solution, the solution
appears green. The color slowly fades to produces a yellow solution, passing
through an intermediate stage in which the solution is red. The green color reappears when
the flask is shaken and then fades to red, then yellow again.
Explanations (including important chemical equations):
Indigo carmine undergoes a reversible reduction.
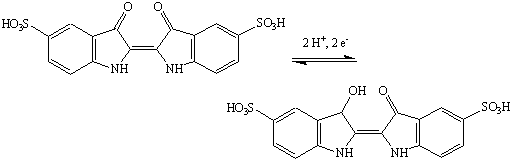
Shaking the flask brings more oxygen into the solution, provoking the formation of the oxidized (green) form. The indigo carmine will eventually undergo reduction again to its reduced (yellow) form. The intermediate color is due to formation of a red semiquinone intermediate.