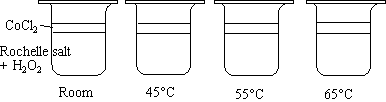
- Four beakers contain equal amounts of rochelle salt solution and 6% H2O2.
- The beakers are heated to their respective temperature.
- CoCl2 is added to each of the four beakers.
Chemical Concepts Demonstrated: Temperature's effect on reactions, heat catalysis
Demonstration:
|
 |
Observations:
The beakers all change color from pink to green when the CoCl2 is added. The solutions turn pink again, but at different times. Typically, the beaker at 25o C changes in 10 minutes, the beaker at 45o C changes in 3 minutes, the beaker at 55o C changes in 1 minute, and the beaker at 65o C quickly flashes through the colors.
Explanation:
Heat itself makes a fairly good catalyst in certain cases. This demonstration can show this fact quite well.
CoCl2, in the presence of water, forms Co(H2O)62+, which is pink. In the presence of both H2O2 and tartrate ion, a green cobalt-tartrate-peroxide complex is formed, which can formally be assumed to contain Co(III). With time, the Co(III) oxidizes one of the tartrate ligands to CO2 and is itself reduced to Co2+; at this point, the pink color of the solution returns. The rate at which the pink color returns depends on the temperature.