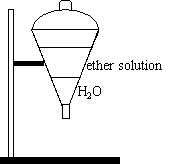
A blue ether solution, made up of KSCN, CoCl2, and ethyl ether, is added to the funnel.
The funnel is stoppered and the contents inside are shaken.
Chemical Concept Demonstrated: The tendency for ionic substrates to extract into more polar solvents
Demonstration:
| 75 mL of water are poured into a funnel. A blue ether solution, made up of KSCN, CoCl2, and ethyl ether, is added to the funnel. The funnel is stoppered and the contents inside are shaken. |
 |
Observations:
The water layer changes into a pink solution at the bottom of the
separatory funnel.
Explanation:
Water is more polar than ethyl ether. Some of the Co2+ ions are extracted into the water layer from the Co(SCN)4 2- complex ions formed in the ether solution. The presence of Cl - can be seen by adding silver nitrate (AgNO3) solution until a white precipitate forms in the water layer at the bottom of the separatory funnel.