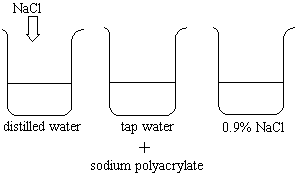
- distilled water
- tap water
- 0.9% NaCl solution
NaCl is added to the first beaker.
Chemical Concept Demonstrated: Ionic strength's effect on the ability of superabsorbent polymers to absorb liquids
Demonstration:
Sodium polyacrylate is added to beakers containing:
NaCl is added to the first beaker. |
 |
Observations:
The polymer absorbs all of the distilled water, most of the tap water,
and some of the NaCl solution. If NaCl is added to the first beaker's resulting gel,
water leaves the polymer network.
Explanation:
Sodium polyacrylate is made by polymerizing a mixture of of sodium acrylate and acrylic acid. The difference between the Na+ ion concentration inside the polymer network and the solution in which it is immersed generates an osmotic pressure that is alleviated as water diffuses into the polymer. As expected, the amount of liquid that can be absorbed depends on the ionic strength of the solution. This polymer can absorb 800 times its own weight of distilled water, 300 times its own weight in tap water, but only 60 times its own weight in urine (~0.9% NaCl).