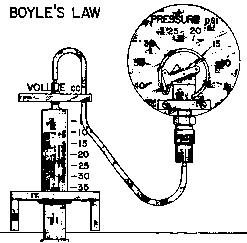
Chemical Concept Demonstrated: Boyle's Law
Demonstration:

| Adjust the plunger so that the syringe contains 30.0 cm3 of air.
Determine the pressure of this gas. Push the plunger in and then let it out to take
volume vs. pressure measurements. |
Observations:
The greater the volume, the lesser the pressure, and vise versa.
If a volume vs. pressure graph is made, a linear relationship can be shown.
Explanation:
There is a fixed number of molecules of air inside of the syringe. When the plunger is pushed in, these molecules begin to push (or collide) against each other and the sides of the syringe more and more because of the lack of space. This decrease in volume (less space) increases the pressure (more collisions). The opposite phenomenon occurs when the plunger is pulled out. Both occurrences are a demonstration of Boyle's Law.