Demonstration:
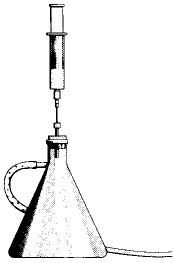
Chemical Concept Demonstrated: Graham's Law of Effusion
Demonstration:

Using the apparatus shown and Graham's Law syringe needles, remove 25 mL of different gases from their balloons.
Then, measure the time it takes for the gases to effuse from the syringe.
Observations:
Three gases were used: hydrogen, oxygen, and difluorodichlormethane.
The hydrogen went the fastest, the oxygen was in the middle, and the
difluorodichloromethane had the slowest rate of effusion.
Explanation:
Graham's law states that the rate of effusion of a gas is inversely propertional to the square root of the density of the gas. Since equal volumes of gas at the same temperature and pressure contain equal numbers of gas molecules, the rate of effusion is also inversely proportional to the square root of the molecular weight of the gas. The gas with the lowest molecular weight effuses the fastest, and vice versa.
It should be noted that Graham's Law of Effusion deals with the rate at which a gas will escape from a pinhole into a vacuum, which is different from Graham's Law of Diffusion (see demonstration 4.19), which deals with the rate at which two gases mix.