- Drop pieces of dry ice into the bottle of water, and close the jar.
- Dip the open end of the PVC pipe in the soap solution.
- Watch as CO2 bubbles form at the end of the pipe.
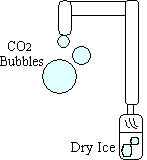
Chemical Concepts Demonstrated: Mass and density of gases
Demonstration:
|
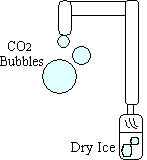 |
Observations:
The bubbles that form on the end of the tube fall to the ground.
Explanation:
It is concievable that students may be under the mistaken impression that gases rise because they have no mass (and, therefore, no density). This demonstration proves that gases do indeed have mass (or, more specifically, differing densities that enable them to rise and sink relative to each other). The carbon dioxide is more dense than the air surrounding it, so bubbles of the gas fall to the ground.