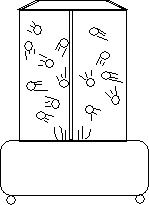
Chemical Concept Demonstrated: Hard-sphere approximation to kinetic molecular theory
| Concept | How the Simulator Demonstrates the Concept |
| The States of Matter |
The simulator can model the differences between the kinetic
energies of gases, liquids, and solids. Simply adjust the speed at which the spheres
are blown about (high speed is gas, low speed is liquid, off is solid). |
| Pressure |
Add a divider and pile all of the spheres to one side.
Note the rate of collosions between the balls and the walls of the container. Removing the
divider increases the volume of the container. Note the change in the rate of collisions. |
| Equilibrium |
Divider now has a 5" hole in the middle. One side
is filled with blue balls, the other side with red balls. Note the difference in the
number of balls on opposite sides of the divider when running the two blowers at the same
power settings, and at different power settings. |
| Entropy |
Once equilibrium has been reached, how long will it take for
all of the red balls to end up on one side again? |
| Activation Energy |
All the balls are now on one side of the divider with the hole
in it. The blower is set on low power and balls are not able to reach the hole. How would
the activation energy be increased in order for the balls to go through the hole? |
| Orientation Between Colliding Particles |
The balls now have velco strips attached to their outsides.
Watch how they interact and begin to form dimers, trimers, tetramers, and even polymer
chains as they collide in the chamber. |
| Evaportation/Diffusion of Gases |
While the balls are rapidly moving in the chamber, the lid is
removed. Watch out for flying "gas!" |
