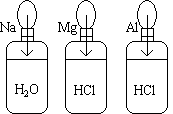9.2 Quantitative and Qualitative Reactions of Active Metals
Chemical Concepts Demonstrated: Reactions of metals, concept of
equivalent weight, stoichiometry
Demonstration:
Three balloons are attached to three bottles as shown in the picture.
- The1st balloon contains a sample of sodium metal and is attached to a bottle
containing water.
- The 2nd balloon contains a sample of magnesium metal and is attached to a
bottle containing HCl.
- The 3rd balloon contains a sample of aluminum metal and is attached to a
bottle containg HCl.
|
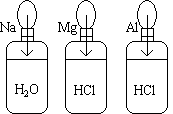 |
The samples are then dropped into their bottles.
Observations:
Although the same amount of metal was used in each trial, the three
balloons contain different volumes of H2 gas. In fact, there is an
approximate 1:2:3 ratio with respect to the resulting sizes of the balloons.
Explanation (including important chemical equations):
2 Na (s) + 2 H2O (l) ---> 1 H2
(g) + 2 NaOH
2 Mg (s) + 4 HCl (l) ----> 2 H2 (g) + 2
MgCl2
2 Al (s) + 6 HCl (l) ----> 3 H2 (g) + 2
AlCl3
The balanced chemical equations show that the stoichiometry of the
three reactions is different. The molar ratios of metal to hydrogen gas indicate that the
aluminum metal will produce the most gas, then magnesium, and finally sodium.
Please note that the movie included is from demonstration 9.3 and is
only useful for the purpose of demonstrating the usage of the equipment.