
Practice Problem 10
Which of the following compounds would form enantiomers because the molecule is chiral?

Solution
The second carbon atom in 2-bromo-2-methylbutane contains two identical CH3 substituents. As a result, this compound is achiral and does not form enantiomers.
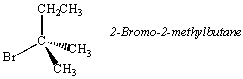
The second carbon atom in 1-bromo-2-methylbutane carries four different substituents: H, Br, CH3, and CH2CH3. As a result, this molecule is chiral and it forms enantiomers.

Note: Every object ![]() with the possible exception of the vampires found on
late-night TV
with the possible exception of the vampires found on
late-night TV ![]() has a mirror image. The relevant question is whether the mirror
image can be superimposed on the source of the image. If it can,
the object is not chiral (achiral). If it cannot, the
object is chiral and it can exist as a pair of stereoisomers.
has a mirror image. The relevant question is whether the mirror
image can be superimposed on the source of the image. If it can,
the object is not chiral (achiral). If it cannot, the
object is chiral and it can exist as a pair of stereoisomers.