
Acids and Bases
For more than 300 years, substances that behaved like vinegar have been classified as acids, while those that have properties like the ash from a wood fire have been called alkalies or bases. The name "acid" comes from the Latin acidus, which means "sour," and refers to the sharp odor and sour taste of many acids. Vinegar tastes sour because it is a dilute solution of acetic acid in water; lemon juice is sour because it contains citric acid; milk turns sour when it spoils because of the formation of lactic acid; and the sour odor of rotten meat can be attributed to carboxylic acids such as butyric acid formed when fat spoils.
Today, when chemists use the words "acid" or "base" they refer to a model developed independently by Brønsted, Lowry, and Bjerrum. Since the most explicit statement of this theory was contained in the writings of Brønsted, it is most commonly known as the "Brønsted acid-base" theory.
Brønsted argued that all acid-base reactions involve the transfer of an H+ ion, or proton. Water reacts with itself, for example, by transferring an H+ ion from one molecule to another to form an H3O+ ion and an OH- ion.

According to this theory, an acid is a "proton donor" and a base is a "proton acceptor."
Acid-Dissociation Equilibrium Constant
Acids are often divided into categories such as "strong" and "weak." One measure of the strength of an acid is the acid-dissociation equilibrium constant, Ka, for that acid.

When Ka is relatively large, we have a strong acid.
| HCl: | Ka = 1 x 103 |
When it is small, we have a weak acid.
| CH3CO2H: | Ka = 1.8 x 10-5 |
When it is very small, we have a very weak acid.
| H2O: | Ka = 1.8 x 10-16 |
In 1909, S. P. L. Sørenson suggested that the enormous range of concentrations of the H3O+ and OH- ions in aqueous solutions could be compressed into a more manageable set of data by taking advantage of logarithmic mathematics and calculating the pH or pOH of the solution.
| pH | = | - log [H3O+] | |
| pOH | = | - log [OH-] |
The "p" in pH and pOH is an operator that indicates that the negative of the logarithm should be calculated for any quantity to which it is attached. Thus, pKa is the negative of the logarithm of the acid-dissociation equilibrium constant.
| pKa | = | - log Ka |
The only disadvantage of using pKa as a measure of the relative strengths of acids is the fact that large numbers now describe weak acids, and small (negative) numbers describe strong acids.
| HCl: | pKa = -3 | ||
| CH3CO2H: | pKa = 4.7 | ||
| H2O: | pKa = 15.7 |
An important features of the Brønsted theory is the relationship it creates between acids and bases. Every Brønsted acid has a conjugate base, and vice versa.


Just as the magnitude of Ka is a measure of the strength of an acid, the value of Kb reflects the strength of its conjugate base. Consider what happens when we multiply the Ka expression for a generic acid (HA) by the Kb expression for its conjugate base (A-).

If we now replace each term in this equation by the appropriate equilibrium constant, we get the following equation.
KaKb = Kw = 1 x 10-14
Because the product of Ka times Kb is a relatively small number, either the acid or its conjugate base can be "strong." But if one is strong, the other must be weak. Thus, a strong acid must have a weak conjugate base.
![]()
A strong base, on the other hand, must have a weak conjugate acid.
![]()
Brønsted Acids and Bases in Nonaqueous Solutions
Water has a limiting effect on the strength of acids and
bases. All strong acids behave the same in water ![]() 1 M
solutions of the strong acids all behave as 1 M
solutions of the H3O+ ion
1 M
solutions of the strong acids all behave as 1 M
solutions of the H3O+ ion ![]() and very
weak acids cannot act as acids in water. Acid-base reactions
don't have to occur in water, however. When other solvents are
used, the full range of acid-base strength shown in the table
below can be observed.
and very
weak acids cannot act as acids in water. Acid-base reactions
don't have to occur in water, however. When other solvents are
used, the full range of acid-base strength shown in the table
below can be observed.
Typical Brønsted Acids and Their Conjugate Bases
| Compound | Ka | pKa | Conjugate Base |
Kb | pKb | |||||
| HI | 3 x 109 | -9.5 | I- | 3 x 10-24 | 23.5 | |||||
| HCl | 1 x 106 | -6 | Cl- | 1 x 10-20 | 20 | |||||
| H2SO4 | 1 x 103 | -3 | HSO4- | 1 x 10-17 | 17 | |||||
| H3O+ | 55 | -1.7 | H2O | 1.8 x 10-16 | 15.7 | |||||
| HNO3 | 28 | -1.4 | NO3- | 3.6 x 10-16 | 15.4 | |||||
| H3PO4 | 7.1 x 10-3 | 2.1 | H2PO4- | 1.4 x 10-12 | 11.9 | |||||
| CH3CO2H | 1.8 x 10-5 | 4.7 | CH3CO2- | 5.6 x 10-10 | 9.3 | |||||
| H2S | 1.0 x 10-7 | 7.0 | HS- | 1 x 10-7 | 7.0 | |||||
| H2O | 1.8 x 10-16 | 15.7 | OH- | 55 | -1.7 | |||||
| CH3OH | 1 x 10-18 | 18 | CH3O- | 1 x 104 | -4 | |||||
| HC |
1 x 10-25 | 25 | HC |
1 x 1011 | -11 | |||||
| NH3 | 1 x 10-33 | 33 | NH2- | 1 x 1019 | -19 | |||||
| H2 | 1 x 10-35 | 35 | H- | 1 x 1021 | -21 | |||||
| CH2=CH2 | 1 x 10-44 | 44 | CH2=CH- | 1 x 1030 | -30 | |||||
| CH4 | 1 x 10-49 | 49 | CH3- | 1 x 1035 | -35 |
The strongest acids are in the upper-left corner of this table; the strongest bases in the bottom-right corner. Each base is strong enough to deprotonate the acid in any line above it. The hydride ion (H-), for example, can convert an alcohol into its conjugate base
![]()
and the amide (NH2-) ion can deprotonate an alkyne.
![]()
In 1923, G. N. Lewis introduced a theory of acids and bases that is even more powerful than the Brønsted theory. As a result, it is important to differentiate between the terms "acid" and "base" as they have been used so far and the terms "Lewis acid" and "Lewis base."
Lewis noted that the Brønsted theory was limited because it focused exclusively on the transfer of a proton (H+). He argued that a more general definition of acid-base reactions could be obtained by looking at what happens when an H+ ion combines with an OH- ion to form water.
![]()
Lewis argued that the H+ ion picks up (or accepts) a pair of electrons from the OH- ion to form a new covalent bond. As a result, any substance that can act as an electron-pair acceptor is a Lewis acid.
Lewis acid: An electron-pair acceptor, such as the H+ ion
The pair of electrons that went into the new covalent bond were donated by the OH- ion. Lewis therefore argued that any substance that can act as an electron-pair donor is a Lewis base.
Lewis base: An electron-pair donor, such as the OH- ion
The Lewis acid-base theory doesn't affect the category of compounds we have called "bases" because any Brønsted base must have a pair of nonbonding electrons in order to accept a proton. This theory, however, vastly expanded the family of compounds that can be called "acids." Anything that has one or more empty valence-shell orbitals can now act as an acid
This theory explains why BF3 reacts instantaneously with NH3. The nonbonding electrons on the nitrogen in ammonia are donated into an empty orbital on the boron to form a new covalent bond, as shown in the figure below.

It also explains why Cu2+ ions pick up ammonia to form the four-coordinate Cu(NH3)42+ ion.
Cu2+(aq) + 4 NH3(aq)![]() Cu(NH3)42+(aq)
Cu(NH3)42+(aq)
In this case, a pair of nonbonding electrons from each of the
four NH3 molecules is donated into an empty orbital on
the Cu2+ ion to form a covalent Cu![]() N bond.
N bond.

The flow of electrons from a Lewis base to a Lewis acid is
often indicated with a curved arrow. The arrow starts on a pair
of nonbonding electrons on the Lewis base and points toward the
Lewis acid with which it reacts. Because adding a pair of
electrons to one point on a molecule often displaces electrons in
the molecule, combinations of curved arrows are often used to
describe even simple chemical reactions. Consider the following
example, in which a pair of electrons on an NH2-
ion are donated to the H+ ion formed when the
electrons in one of the C![]() H bonds in acetylene are given to the
carbon atom instead of being shared by the C and H atoms in this
bond.
H bonds in acetylene are given to the
carbon atom instead of being shared by the C and H atoms in this
bond.

Using Acids and Bases to Understand Grignard Reagents
The discussion of acids and bases in the previous section helps us understand the chemistry of the Grignard reagents. Grignard reagents are made by reacting an alkyl bromide with magnesium metal in diethyl ether.

An analogous reagent, known as an alkyllithium, can be prepared by reacting the alkyl bromide with lithium metal in diethyl ether.
![]()
In the course of these reactions the carbon atom is reduced from the -2 to the -4 oxidation state. Whereas the starting material contains a carbon atom with a partial positive charge, the carbon atom in the products of these reactions carries a partial negative charge.

CH3Li and CH3MgBr can therefore be thought of as a source of the CH3- ion.
![]()
The CH3- ion is the conjugate base of methane, which is the weakest Brønsted acid in the table of Brønsted acids and their conjugate bases.
![]()
The CH3- ion is therefore the strongest Brønsted base in this table.
A graduate student once tried to run the following reaction to prepare a Grignard reagent. Explain what he did wrong, why the yield of the desired product was zero, and predict the product he obtained.
| Mg | ||
| CH3CH2Br | CH3CH2MgBr | |
| CH3CH2OH |
Click here to check your answer to practice problem 1
Carbanion Attack at a Carbonyl Group
A subtle, but important, point must be made before we can extend our understanding of acid-base chemistry to the reaction between a Grignard or alkyllithium reagent and a carbonyl group. The data in the table of Brønsted acids and their conjugate bases reflect the strengths of common acids and bases when they act as Brønsted acids or bases. These data predict that methyllithium should react with acetylene to form methane and an acetylide ion, for example.

This reaction should occur because it converts the stronger of a pair of Brønsted acids and the stronger of a pair of Brønsted bases into a weaker acid and a weaker base.

The reaction between a carbonyl and CH3Li or CH3MgBr, on the other hand, involves attack by a CH3- ion acting as a Lewis base or nucleophile at the positive end of the carbonyl group.

This raises an interesting question: Is the stronger of a pair of Brønsted bases always the stronger of a pair of Lewis bases? Unfortunately, the answer is no, it isn't. At times, the stronger of a pair of Brønsted bases is the weaker Lewis base or nucleophile. As a rule, however, strong Brønsted bases are strong nucleophiles, and weak Brønsted bases are weak nucleophiles.
Despite the enormous utility of the Grignard reagent in organic chemistry, the exact mechanism of the reaction between these reagents and a carbonyl is not known. There is reason to believe that two molecules of the Grignard reagent are involved in this reaction. The magnesium atom of one molecule of this reagent acts as a Lewis acid that interacts with the oxygen atom of the carbonyl group. The alkyl group of the other reagent then acts as a Lewis base, attacking the positive end of the carbonyl.

In essence, this reaction involves the attack by a negatively
charged CH3- ion at the positively charged
end of the carbonyl group. When this happens, the pair of
nonbonding electrons on the CH3- ion are
used to from a C![]() C bond. This, in turn, displaces the pair of electrons
in the bond onto the other end of the carbonyl group.
C bond. This, in turn, displaces the pair of electrons
in the bond onto the other end of the carbonyl group.
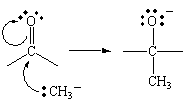
The second molecule of the Grignard reagent, which binds at the oxygen end of the carbonyl, isn't consumed in the reaction. Its function is simple. When it acts as a Lewis acid, binding to the oxygen atom in the C=O double, it increases the polarity of this bond. By making the bond more polar, it increases the rate at which the CH3- ion attacks the positive end of the C=O bond.