The Chemistry Of Nonmetals
More than 75% of the known elements have the characteristic properties of metals (see figure below). They have a metallic luster; they are malleable and ductile; and they conduct heat and electricity. Eight other elements (B, Si, Ge, As, Sb, Te, Po, and At) are best described as semimetals or metalloids. They often look like metals, but they tend to be brittle, and they are more likely to be semiconductors than conductors of electricity.
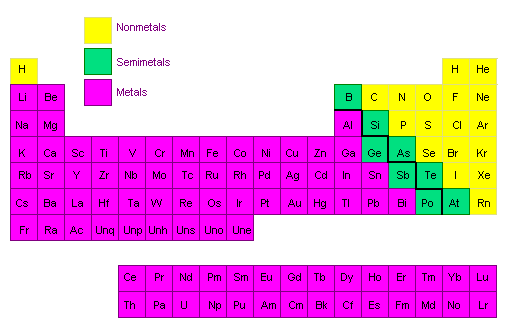 |
Once the metals and semimetals are removed from the list of known elements, only 17 are left to be classified as nonmetals. Six of these elements belong to the family of rare gases in Group VIIIA, most of which are virtually inert to chemical reactions. Discussions of the chemistry of the nonmetals therefore tend to focus on the following elements: H, C, N, O, F, P, S, Cl, Se, Br, I, and Xe.
There is a clear pattern in the chemistry of the main group metals: The main group metals are oxidized in all of their chemical reactions. These metals are oxidized when they react with nonmetal elements. Aluminum, for example, is oxidized by bromine.

The chemistry of the nonmetals is more interesting because these elements can undergo both oxidation and reduction. Phosphorus, for example, is oxidized when it reacts with oxygen to form P4O10.

But it is reduced when it reacts with calcium to form calcium phosphide.

These reactions can be understood by looking at the relative electronegativities of these elements. Phosphorus (EN = 2.19) is less electronegative than oxygen (EN = 3.44). When these elements react, the electrons are drawn toward the more electronegative oxygen atoms. Phosphorus is therefore oxidized in this reaction, and oxygen is reduced. Calcium (EN = 1.00), on the other hand, is significantly less electronegative than phosphorus (EN = 2.19). When these elements react, the electrons are drawn toward the more electronegative phosphorus atoms. As a result, calcium is oxidized and phosphorus is reduced.
The behavior of the nonmetals can be summarized as follows.
1. Nonmetals tend to oxidize metals.
| 2 Mg(s) | + | O2(g) | 2 MgO(s) |
2. Nonmetals with relatively large electronegativities (such as oxygen and chlorine) oxidize substances with which they react.
| 2 H2S(g) | + | 3 O2(g) | 2 SO2(g) | + | 2 H2O(g) |
| PH3(g) | + | 3 Cl2(g) | PCl3(l) | + | 3 HCl(g) |
3. Nonmetals with relatively small electronegativities (such as carbon and hydrogen) can reduce other substances.
| Fe2O3(s) | + | 3 C(s) | 2 Fe(s) | + | 3 CO(g) |
| CuO(s) | + | H2(g) | Cu(s) | + | H2O(g) |
| Practice Problem 1: Determine which element is oxidized and which is reduced when sulfur vapor reacts with red-hot charcoal to form carbon disulfide. 4 C(s) + S8(g)
|
