One of the most important![]() and time-consuming
and time-consuming ![]() activities
in chemistry involves isolating, separating, and purifying
chemical compounds. Extraction (literally,
"taking out by force") is a useful technique for
separating compounds such as I2 and KMnO4
that have different polarities. The compounds to be separated are
treated with a mixture of a polar solvent (such as H2O)
and a nonpolar solvent (such as CCl4). The I2
will dissolve in the CCl4, while the KMnO4
will dissolve in the H2O. By separating these two
phases and allowing the solvents to evaporate, we can cleanly
separate I2 and KMnO4.
activities
in chemistry involves isolating, separating, and purifying
chemical compounds. Extraction (literally,
"taking out by force") is a useful technique for
separating compounds such as I2 and KMnO4
that have different polarities. The compounds to be separated are
treated with a mixture of a polar solvent (such as H2O)
and a nonpolar solvent (such as CCl4). The I2
will dissolve in the CCl4, while the KMnO4
will dissolve in the H2O. By separating these two
phases and allowing the solvents to evaporate, we can cleanly
separate I2 and KMnO4.
This technique can also be used to extract solutes from a solid. Coffee, for example, is decaffeinated by extraction. Most of the popularity of coffee can be attributed to the stimulating effect of caffeine, which makes up as much as 2.5% of the dry weight of coffee beans. But many people who have grown to like the flavor of coffee want to reduce the amount of caffeine they consume and therefore turn to decaffeinated coffee. Caffeine can be extracted with hot water, but this also tends to extract the oils that give coffee its flavor. Coffee beans were therefore decaffeinated for many years by treating them with a nonpolar solvent such as dichloromethane (CH2Cl2), which dissolves most of the caffeine without destroying the flavor of the coffee. (The caffeine is then sold to manufacturers of cola drinks, who add it to their products.)
Distillation is the technique used most frequently to purify liquids. At its simplest, it involves heating the liquid, as shown in the figure below, until it boils. The vapor that escapes is passed through a water-cooled condenser, where it condenses to form a liquid, which collects in a clean flask.
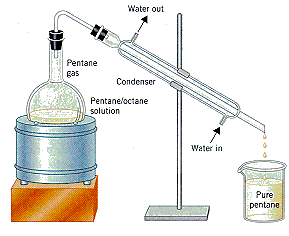 |
| A distillation apparatus |
| Practice Problem 10: Assume that a mixture that contains equal amounts of pentane (C5H12) and octane (C8H18) is distilled. Describe the difference between the composition of the liquid in the distillation flask and the vapor given off when this liquid starts to boil. |
We can develop a general rule for distillations by constructing a phase diagram that describes the mixtures under all possible combinations of temperature and percent composition, as shown in the figure below.
 |
| A phase diagram for a mixture of pentane and octane that shows the effect of changes in the composition of the mixture on the boiling point of the solution. |
The points labeled Tp and To in this diagram represent the boiling points of pure pentane and pure octane. These points are connected by two curves. At temperatures below the bottom curve, mixtures of pentane and octane are liquids. At temperatures above the top curve, these mixtures are gases. Points that lie between the curves describe systems that contain both a liquid and a gas phase with the same composition. The liquid and the gas should not have the same composition at a given temperature. Points between the two curves are therefore unstable. Any system at point B, for example, would come to equilibrium by forming a vapor with the composition described by point A and a liquid with the composition described by point C.
The vertical line in the figure above shows what happens to the system when a 50 : 50 mixture of pentane and octane is heated. Nothing changes until the temperature reaches point C -- the temperature at which the liquid starts to boil. The vapor that collects above the boiling liquid will have the composition described by point A in this diagram. It will therefore be much richer in pentane than the liquid from which it boils. If this vapor escapes, more pentane leaves the system than octane, and the percent by mass of octane in the system increases. This increases the boiling point of the mixture, as well as gradually increasing the amount of octane that collects in the vapor that escapes from the liquid.
This phase diagram therefore provides the basis for separating a mixture by distillation. We start by heating the mixture and collecting the first few portions, or fractions, that distill over. These fractions will be rich in the component that has the lower boiling point. We might then discard the next few fractions, which contain a mixture of the low- and high-boiling compounds, and save the last few fractions, which should be rich in the compound that has the higher boiling point.
Distillation works best for purifying compounds that are liquids at room temperature. Solids are more likely to be purified by recrystallization, which takes advantage of differences in the solubility of various components of a mixture in different solvents. We start by dissolving as much of the solid as possible in a hot sample of the solvent. We then cool the solution until some of the solid crystallizes back out of solution. Each time the sample is dissolved and recrystallized, it is purer than before. This was the technique used by Marie and Pierre Curie to isolate the first minuscule sample of radium from several tons of the mineral pitchblende.
One of the fastest-growing methods of separating compounds was developed by a Russian botanist named Mikhail Tsvet in 1906. This technique was called chromatography (literally, "writing with color") because it was first used to separate the colored pigments in plants. The basic principle behind chromatography is simple. A gas or liquid is allowed to flow over a solid support. Compounds that have a high affinity for the solvent are carried along as it moves past the stationary support. Compounds that have a higher affinity for the solid move more slowly.
In thin-layer chromatography (TLC), a glass plate or plastic support is coated with a thin layer of a solid such as alumina (Al2O3) or silica (SiO2). Small samples of the compounds to be separated are then placed on the silica or alumina, and the TLC plate is immersed in a solvent until the solvent line is just below the point where the samples were applied (see figure below). The solvent slowly moves up the plate, carrying the components of the mixture with it. Some of these components have a high affinity for the Al2O2 or SiO2 support, and they move very slowly. Others have a high affinity for the solvent, and they move more rapidly. The net result is a separation of the mixture into its components based on their relative affinities for the stationary solid phase and the mobile liquid phase.
 |
| Thin-layer chromatography (TLC) (a) The solution is applied near one end of a plate coated with silica or alumina. (b) The plate is then immersed in a solvent, which rises up the plate by capillary action. |
Column chromatography extends the principles of TLC to the large-scale separation of mixtures. This technique involves filling a glass column with a solid support, applying up to several grams of the mixture to the top of the column, and then slowly washing the column with solvent.
Mixtures can also be separated by gas-phase chromatography, which takes advantage of the relative affinity of the different components in a mixture for the stationary support when the mixture is heated until there is an equilibrium between its gas and liquid phases.
Zone refining is used to obtain very pure (99.999%) samples of metals and semimetals. The starting material is drawn into a thin rod which is inserted into a heating coil that slowly moves along the length of the rod. As the coil moves, it constantly brings a new portion of the rod to the point at which the solid just melts. Because solutions melt at lower temperatures than pure solids, it is possible to concentrate the impurities in the small sample of liquid that moves just ahead of the heating coil, and crystallize almost perfectly pure metal or semimetal just behind the coil.
