| The Activity of Metals | Classifying Metals Based on Activity |
| Predicting the Product of Main Group Metal Reactions | |
The primary difference between metals is the ease with which they undergo chemical reactions. The elements toward the bottom left corner of the periodic table are the metals that are the most active in the sense of being the most reactive. Lithium, sodium, and potassium all react with water, for example. The rate of this reaction increases as we go down this column, however, because these elements become more active as they become more metallic.
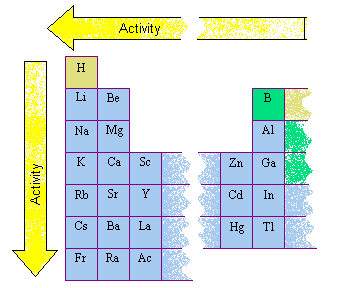
Classifying Metals Based on Activity
The metals are often divided into four classes on the basis of their activity, as shown in the table below.
Common Metals Divided into Classes on the Basis of Their Activity
| Class I Metals: The Active Metals |
| Li, Na, K, Rb, Cs (Group IA) |
| Ca, Sr, Ba (Group IIA) |
| Class II Metals: The Less Active Metals |
| Mg, Al, Zn, Mn |
| Class III Metals: The Structural Metals |
| Cr, Fe, Sn, Pb, Cu |
| Class IV Metals: The Coinage Metals |
| Ag, Au, Pt, Hg |
The most active metals are so reactive that they readily combine with the O2 and H2O vapor in the atmosphere and are therefore stored under an inert liquid, such as mineral oil. These metals are found exclusively in Groups IA and IIA of the periodic table.
Metals in the second class are slightly less active. They don't react with water at room temperature, but they react rapidly with acids.
The third class contains metals such as chromium, iron, tin, and lead, which react only with strong acids. It also contains even less active metals such as copper, which only dissolves when treated with acids that can oxidize the metal.
Metals in the fourth class are so unreactive they are essentially inert at room temperature. These metals are ideal for making jewelry or coins because they do not react with the vast majority of the substances with which they come into daily contact. As a result, they are often called the "coinage metals."
Predicting the Product of Main Group Metal Reactions
The product of many reactions between main group metals and other elements can be predicted from the electron configurations of the elements.
Example: Consider the reaction between sodium and chlorine to form sodium chloride. It takes more energy to remove an electron from a sodium atom to form an Na+ ion than we get back when this electron is added to a chlorine atom to form a Cl- ion. Once these ions are formed, however, the force of attraction between these ions liberates enough energy to make the following reaction exothermic.
| Na(s) + 1/2
Cl2(g) |
The net effect of this reaction is to transfer one electron from a neutral sodium atom to a neutral chlorine atom to form Na+ and Cl- ions that have filled-shell configurations.
![]()
Potassium and hydrogen have the following electron configurations.
| K: [Ar] 4s1 | H: 1s1 |
When these elements react, an electron has to be transferred from one element to the other. We can decide which element should lose an electron by comparing the first ionization energy for potassium (418.8 kJ/mol) with that for hydrogen (1312.0 kJ/mol).
Potassium is much more likely to lose an electron in this reaction, which means that hydrogen gains an electron to form K+ and H- ions.
![]()
| Practice Problem 1: Write a balanced equation for the following reaction. Li(s) + O2(s)
|
