The Reactions of Alkanes, Alkenes, and Alkynes
In the absence of a spark or a high-intensity light source, alkanes are generally inert to chemical reactions. However, anyone who has used a match to light a gas burner, or dropped a match onto charcoal coated with lighter fluid, should recognize that alkanes burst into flame in the presence of a spark. It doesn't matter whether the starting material is the methane found in natural gas,
CH4(g) + 2 O2(g) ![]() CO2(g)
+ 2 H2O(g)
CO2(g)
+ 2 H2O(g)
the mixture of butane and isobutane used in disposable cigarette lighters,
2 C4H10(g) + 13 O2(g)
![]() 8 CO2(g)
+ 10 H2O(g)
8 CO2(g)
+ 10 H2O(g)
the mixture of C5 to C6 hydrocarbons in charcoal lighter fluid,
C5H12(g) + 8 O2(g) ![]() 5 CO2(g)
+ 6 H2O(g)
5 CO2(g)
+ 6 H2O(g)
or the complex mixture of C6 to C8 hydrocarbons in gasoline.
2 C8H18(l) + 25 O2(g)
![]() 16 CO2(g)
+ 18 H2O(g)
16 CO2(g)
+ 18 H2O(g)
Once the reaction is ignited by a spark, these hydrocarbons burn to form CO2 and H2O and give off between 45 and 50 kJ of energy per gram of fuel consumed.
In the presence of light, or at high temperatures, alkanes react with halogens to form alkyl halides. Reaction with chlorine gives an alkyl chloride.
| light | ||
| CH4(g) + Cl2(g) | CH3Cl(g) + HCl(g) |
Reaction with bromine gives an alkyl bromide.
| light | ||
| CH4(g) + Br2(l) | CH3Br(g) + HBr(g) |
Unsaturated hydrocarbons such as alkenes and alkynes are much more reactive than the parent alkanes. They react rapidly with bromine, for example, to add a Br2 molecule across the C=C double bond.
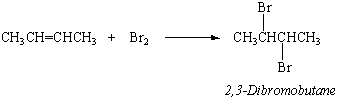
This reaction provides a way to test for alkenes or alkynes. Solutions of bromine in CCl4 have an intense red-orange color. When Br2 in CCl4 is mixed with a sample of an alkane, no change is initially observed. When it is mixed with an alkene or alkyne, the color of Br2 rapidly disappears.
The reaction between 2-butene and bromine to form 2,3-dibromobutane is just one example of the addition reactions of alkenes and alkynes. Hydrogen bromide (HBr) adds across a C=C double bond to form the corresponding alkyl bromide, in which the hydrogen ends up on the carbon atom that had more hydrogen atoms to begin with. Addition of HBr to 2-butene, for example, gives 2-bromobutane.

H2 adds across double (or triple bonds) in the presence of a suitable catalyst to convert an alkene (or alkyne) to the corresponding alkane.
![]()
In the presence of an acid catalyst, it is even possible to add a molecule of water across a C=C double bond.

Addition reactions provide a way to add new substituents to a hydrocarbon chain and thereby produce new derivatives of the parent alkanes.
In theory, two products can form when an unsymmetric reagent such as HBr is added to an unsymmetric C=C double bond. In practice, only one product is obtained. When HBr is added to 2-methylpropene, for example, the product is 2-bromo-2-methylpropane, not 1-bromo-2-methylpropane.

In 1870, after careful study of many examples of addition reactions, the
Russian chemist Valdimir Markovnikov formulated a rule for predicting the product of these
reactions. Markovnikov's rule states that the hydrogen atom adds to the carbon
atom that already has the larger number of hydrogen atoms when HX adds to an alkene.
Thus, water (H![]() OH) adds to propene to
form the product in which the OH group is on the middle carbon atom.
OH) adds to propene to
form the product in which the OH group is on the middle carbon atom.

Organic Chemistry: Structure and Nomenclature of Hydrocarbons
Structure and Nomenclature of Hydrocarbons | Isomers | The Reactions of Alkanes, Alkenes, and Alkynes | Hydrocarbons | Petroleum and Coal | Chirality and Optical Activity
Periodic Table | Periodic Table | Glossary | Cool Applets
Gen Chem Topic Review | General Chemistry Help Homepage | Search: The general chemistry web site.