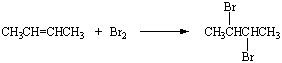
Functional Groups
Bromine reacts with 2-butene to form 2,3-dibromobutane.
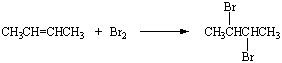
It also reacts with 3-methyl-2-pentene to form 2,3-dibromopentane.
Instead of trying to memorize both equations, we can build a general rule that bromine reacts with compounds that contain a C=C double bond to give the product expected from addition across the double bond. This approach to understanding the chemistry of organic compounds presumes that certain atoms or groups of atoms known as functional groups give these compounds their characteristic properties.
Functional groups focus attention on the important aspects of the structure of a molecule. We don't have to worry about the differences between the structures of 1-butene and 2-methyl-2-hexene, for example, when these compounds react with hydrogen bromide. We can focus on the fact that both compounds are alkenes that add HBr across the C=C double bond in the direction predicted by Markovnikov's rule.
Some common functional groups are given in the table below.
Common Functional Groups
| Functional Group | Name | Example | ||
 |
Alkane | CH3CH2CH3 (propane) | ||
| Alkene | CH3CH=CH2 (propene) | |||
| Alkyne | CH3C |
|||
| F, Cl, Br, or I | Alkyl halide | CH3Br (methyl bromide) | ||
| Alcohol | CH3CH2OH (ethanol) | |||
| Ether | CH3OCH3 (dimethyl ether) | |||
| Amine | CH3NH2 (methyl amine) |
The C=O group plays a particularly important role in organic chemistry. This group is called a carbonyl and some of the functional groups based on a carbonyl are shown in the table below.
Functional Groups That Contain a Carbonyl
| Practice Problem 1: Root beer hasn't tasted the same since the use of sassafras oil as a food additive was outlawed because sassafras oil is 80% safrole, which has been shown to cause cancer in rats and mice. Identify the functional groups in the structure of safrole.
|
| Practice Problem 2: The following compounds are the active ingredients in over-the-counter drugs used as analgesics (to relieve pain without decreasing sensibility or consciousness), antipyretics (to reduce the body temperature when it is elevated), and/or anti-inflammatory agents (to counteract swelling or inflammation of the joints, skin, and eyes). Identify the functional groups in each molecule.
|
| Practice Problem 3: The discovery of penicillin in 1928 marked the beginning of what has been called the "golden age of chemotherapy," in which previously life-threatening bacterial infections were transformed into little more than a source of discomfort. For those who are allergic to penicillin, a variety of antibiotics, including tetracycline, are available. Identify the numerous functional groups in the tetracycline molecule.
|
Focusing on the functional groups in a molecule allows us to recognize patterns in the behavior of related compounds. Consider what we know about the reaction between sodium metal and water, for example.
2 Na(s) + 2 H2O(l) ![]() H2(g)
+ 2 Na+(aq) + 2 OH-(aq)
H2(g)
+ 2 Na+(aq) + 2 OH-(aq)
We can divide this reaction into two half-reactions. One involves the oxidation of sodium metal to form sodium ions.
| Oxidation: | Na | Na+ + e- |
The other involves the reduction of an H+ ion in water to form a neutral hydrogen atom that combines with another hydrogen atom to form an H2 molecule.
| Reduction: |  |
Once we recognize that water contains an![]() OH functional group, we can predict what might happen when sodium metal reacts
with an alcohol that contains the same functional group. Sodium metal should react with
methanol (CH3OH), for example, to give H2 gas and a solution of the
Na+ and CH3O- ions dissolved in this alcohol.
OH functional group, we can predict what might happen when sodium metal reacts
with an alcohol that contains the same functional group. Sodium metal should react with
methanol (CH3OH), for example, to give H2 gas and a solution of the
Na+ and CH3O- ions dissolved in this alcohol.
2 Na(s) + 2 CH3OH(l) ![]() H2(g)
+ 2 Na+(alc) + 2 CH3O-(alc)
H2(g)
+ 2 Na+(alc) + 2 CH3O-(alc)
Because they involve the transfer of electrons, the reaction between sodium metal and either water or an alcohol are examples of oxidation-reduction reactions. But what about the following reaction, in which hydrogen gas reacts with an alkene in the presence of a transition metal catalyst to form an alkane?
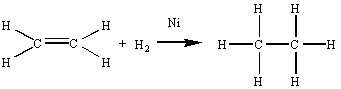
There is no change in the number of valence electrons on any of the atoms in this
reaction. Both before and after the reaction, each carbon atom shares a total of eight
valence electrons and each hydrogen atom shares two electrons. Instead of electrons, this
reaction involves the transfer of atoms![]() in this case, hydrogen atoms. There are so many atom-transfer reactions that
chemists developed the concept of oxidation number to extend the idea of
oxidation and reduction to reactions in which electrons aren't necessarily gained or lost.
in this case, hydrogen atoms. There are so many atom-transfer reactions that
chemists developed the concept of oxidation number to extend the idea of
oxidation and reduction to reactions in which electrons aren't necessarily gained or lost.
| Oxidation involves an increase in the oxidation number of an atom. |
| Reduction occurs when the oxidation number of an atom decreases. |
During the transformation of ethene into ethane, there is a decrease in the oxidation number of the carbon atom. This reaction therefore involves the reduction of ethene to ethane.
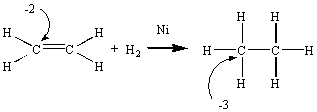
Reactions in which none of the atoms undergo a change in oxidation number are called metathesis reactions. Consider the reaction between a carboxylic acid and an amine, for example.
![]()
Or the reaction between an alcohol and hydrogen bromide.
![]()
These are metathesis reactions because there is no change in the oxidation number of any atom in either reaction.
The oxidation numbers of the carbon atoms in a variety of compounds are given in the table below.
Typical Oxidation Numbers of Carbon
| Functional Group | Example | Oxidation Number of Carbon in the Example |
||
| Alkane | CH4 | -4 | ||
| Alkyllithium | CH3Li | -4 | ||
| Alkene | H2C=CH2 | -2 | ||
| Alcohol | CH3OH | -2 | ||
| Ether | CH3OCH3 | -2 | ||
| Alkyl halide | CH3Cl | -2 | ||
| Amine | CH3NH2 | -2 | ||
| Alkyne | HC |
-1 | ||
| Aldehyde | H2CO | 0 | ||
| Carboxylic acid | HCO2H | 2 | ||
| CO2 | 4 |
These oxidation numbers can be used to classify organic reactions as either oxidation-reduction reactions or metathesis reactions.
| Practice Problem 4: Classify the following as either oxidation-reduction or metathesis reactions.
|
Because electrons are neither created nor destroyed, oxidation can't occur in the absence of reduction, or vice versa. It is often useful, however, to focus attention on one component of the reaction and ask: Is that substance oxidized or reduced?
| Practice Problem 5: Determine whether the following transformations involve the oxidation or the reduction of the carbon atom.
|
Assigning oxidation numbers to the individual carbon atoms in a complex molecule can be difficult. Fortunately, there is another way to recognize oxidation-reduction reactions in organic chemistry:
Oxidation occurs when hydrogen atoms are removed from a carbon atom or when an oxygen atom is added to a carbon atom.
Reduction occurs when hydrogen atoms are added to a carbon atom or when an oxygen atom is removed from a carbon atom.
The first reaction in practice problem 5 involves oxidation of the carbon atom because a pair of hydrogen atoms are removed from that atom when the alcohol is oxidized to an aldehyde.
![]()
The second reaction in practice problem 5 is an example of oxidation because an oxygen atom is added to the carbon atom when an aldehyde is oxidized to a carboxylic acid.
![]()
Reduction, on the other hand, occurs when hydrogen atoms are added to a carbon atom or when an oxygen atom is removed from a carbon atom. An alkene is reduced, for example, when it reacts with H2 to form the corresponding alkane.
![]()
The figure below provides a useful guide to the oxidation-reduction reactions of organic compounds. Each of the arrows in this figure involves a two-electron oxidation of a carbon atom along the path toward carbon dioxide. A line is drawn through the first arrow because it is impossible to achieve this transformation in a single step.
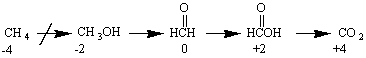
Organic Chemistry: Functional Groups
Functional Groups | Alkyl Halides | Alcohols and Ethers | Aldehydes and Ketones | The Carbonyl Group | Amines, Alkaloids, and Amides | Grignard Reagents
Research in the 1990's: The Chemistry of Garlic
Periodic Table | Glossary | Cool Applets
Gen Chem Topic Review | General Chemistry Help Homepage | Search: The general chemistry web site.