Why Do Some Solids Dissolve in Water?
The sugar we use to sweeten coffee or tea is a molecular solid, in which the individual molecules are held together by relatively weak intermolecular forces. When sugar dissolves in water, the weak bonds between the individual sucrose molecules are broken, and these C12H22O11 molecules are released into solution.
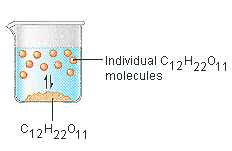
It takes energy to break the bonds between the C12H22O11 molecules in sucrose. It also takes energy to break the hydrogen bonds in water that must be disrupted to insert one of these sucrose molecules into solution. Sugar dissolves in water because energy is given off when the slightly polar sucrose molecules form intermolecular bonds with the polar water molecules. The weak bonds that form between the solute and the solvent compensate for the energy needed to disrupt the structure of both the pure solute and the solvent. In the case of sugar and water, this process works so well that up to 1800 grams of sucrose can dissolve in a liter of water.
Ionic solids (or salts) contain positive and negative ions, which are held together by the strong force of attraction between particles with opposite charges. When one of these solids dissolves in water, the ions that form the solid are released into solution, where they become associated with the polar solvent molecules.

| H2O | ||||
| NaCl(s) | Na+(aq) | + | Cl-(aq) |
We can generally assume that salts dissociate into their ions when they dissolve in water. Ionic compounds dissolve in water if the energy given off when the ions interact with water molecules compensates for the energy needed to break the ionic bonds in the solid and the energy required to separate the water molecules so that the ions can be inserted into solution.
Discussions of solubility equilibria are based on the following assumption: When solids dissolve in water, they dissociate to give the elementary particles from which they are formed. Thus, molecular solids dissociate to give individual molecules
| H2O | ||
| C12H22O11(s) | C12H22O11(aq) |
and ionic solids dissociate to give solutions of the positive and negative ions they contain.
| H2O | ||||
| NaCl(s) | Na+(aq) | + | Cl-(aq) |
When the salt is first added, it dissolves and dissociates rapidly. The conductivity of the solution therefore increases rapidly at first.
| dissolve | ||||
| NaCl(s) | Na+(aq) | + | Cl-(aq) | |
| dissociate |

The concentrations of these ions soon become large enough that the reverse reaction starts to compete with the forward reaction, which leads to a decrease in the rate at which Na+ and Cl- ions enter the solution.
| associate | ||||
| Na+(aq) | + | Cl-(aq) | NaCl(s) | |
| precipitate |
Eventually, the Na+ and Cl- ion concentrations become large enough that the rate at which precipitation occurs exactly balances the rate at which NaCl dissolves. Once that happens, there is no change in the concentration of these ions with time and the reaction is at equilibrium. When this system reaches equilibrium it is called a saturated solution, because it contains the maximum concentration of ions that can exist in equilibrium with the solid salt. The amount of salt that must be added to a given volume of solvent to form a saturated solution is called the solubility of the salt.
There are a number of patterns in the data obtained from measuring the solubility of different salts. These patterns form the basis for the rules outlined in the table below, which can guide predictions of whether a given salt will dissolve in water. These rules are based on the following definitions of the terms soluble, insoluble, and slightly soluble.
- A salt is soluble if it dissolves in water to give a solution with a concentration of at least 0.1 moles per liter at room temperature.
- A salt is insoluble if the concentration of an aqueous solution is less than 0.001 M at room temperature.
- Slightly soluble salts give solutions that fall between these extremes.
Solubility Rules for Ionic Compounds in Water
Soluble Salts1. The Na+, K+, and NH4+ ions form soluble salts. Thus, NaCl, KNO3, (NH4)2SO4, Na2S, and (NH4)2CO3 are soluble. 2. The nitrate (NO3-) ion forms soluble salts. Thus, Cu(NO3)2 and Fe(NO3)3 are soluble. 3. The chloride (Cl-), bromide (Br-), and iodide (I-) ions generally form soluble salts. Exceptions to this rule include salts of the Pb2+, Hg22+, Ag+, and Cu+ ions. ZnCl2 is soluble, but CuBr is not. 4. The sulfate (SO42-) ion generally forms soluble salts. Exceptions include BaSO4, SrSO4, and PbSO4, which are insoluble, and Ag2SO4, CaSO4, and Hg2SO4, which are slightly soluble.
Insoluble Salts1. Sulfides (S2-) are usually insoluble. Exceptions include Na2S, K2S, (NH4)2S, MgS, CaS, SrS, and BaS. 2. Oxides (O2-) are usually insoluble. Exceptions include Na2O, K2O, SrO, and BaO, which are soluble, and CaO, which is slightly soluble. 3. Hydroxides (OH-) are usually insoluble. Exceptions include NaOH, KOH, Sr(OH)2, and Ba(OH)2, which are soluble, and Ca(OH)2, which is slightly soluble. 4. Chromates (CrO42-) are usually insoluble. Exceptions include Na2CrO4, K2CrO4, (NH4)2CrO4, and MgCrO4. 5. Phosphates (PO43-) and carbonates (CO32-) are usually insoluble. Exceptions include salts of the Na+, K+, and NH4+ ions.
