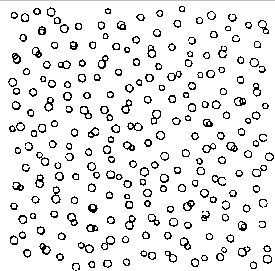
Ceramics
One of the characteristic properties of a substance is its viscosity, which is a measure of its resistance to flow. Motor oils are more viscous than gasoline, for example, and the maple syrup used on pancakes is more viscous than the vegetable oils used in salad dressings. Viscosity depends on any factor that can influence the ease with which molecules slip past each other. Liquids tend to become more viscous as the molecules become larger, or as the intermolecular forces become stronger. They also become more viscous when cooled.
Imagine what would happen if you cooled a liquid until it became so viscous that it was rigid and yet it lacked any of the long-range order that characterizes the solids discussed in this chapter. You would have something known as a glass. Glasses have three characteristics that make them more closely resemble "frozen liquids" than crystalline solids. First, and foremost, there is no long-range order. Second, there are numerous empty sites or vacancies. Finally, glasses don't contain planes of atoms.
The simplest way to understand the difference between a glass and a crystalline solid is to look at the structure of glassy metals at the atomic scale. By rapidly condensing metal atoms from the gas phase, or by rapidly quenching a molten metal, it is possible to produce glassy metals that have the structure shown in the figure below
The structure of a glassy metal on the atomic scale.
The amorphous structure of glass makes it brittle. Because glass doesn't contain planes of atoms that can slip past each other, there is no way to relieve stress. Excessive stress therefore forms a crack that starts at a point where there is a surface flaw. Particles on the surface of the crack become separated. The stress that formed the crack is now borne by particles that have fewer neighbors over which the stress can be distributed. As the crack grows, the intensity of the stress at its tip increases. This allows more bonds to break, and the crack widens until the glass breaks. Thus, if you want to cut a piece of glass, start by scoring the glass with a file to produce a scratch along which it will break when stressed.
Glass has been made for at least 6000 years, since the Egyptians coated figurines made from sand (SiO2) with sediment from the Nile river, heated these objects until the coating was molten, and then let them cool. Calcium oxide or "lime" (CaO) and sodium oxide or "soda" (Na2O) from the sediment flowed into the sand to form a glass on the surface of the figurines. Trace amounts of copper oxide (CuO) in the sediment gave rise to a random distribution of Cu2+ ions in the glass that produced a characteristic blue color.
Sand is still the most common ingredient from which glass is made. (More than 90% of
the sand consumed each year is used by the glass industry.) Sand consists of an irregular
network of silicon atoms held together by Si![]() O
O![]() Si bonds. If the
network was perfectly regular, each silicon atom would be surrounded by four oxygen atoms
arranged toward the corner of a tetrahedron. Because each oxygen atom in this network is
shared by two silicon atoms, the empirical formula of this solid would be SiO2
and the material would have the structure of quartz. In sand, however, some of the Si
Si bonds. If the
network was perfectly regular, each silicon atom would be surrounded by four oxygen atoms
arranged toward the corner of a tetrahedron. Because each oxygen atom in this network is
shared by two silicon atoms, the empirical formula of this solid would be SiO2
and the material would have the structure of quartz. In sand, however, some of the Si![]() O
O![]() Si bridges are broken, in a random fashion.
Si bridges are broken, in a random fashion.
Modifiers (or fluxes) such as Na2O and CaO are added to the sand to alter
the network structure by replacing Si![]() O
O![]() Si bonds with Si
Si bonds with Si![]() O- Na+ or Si
O- Na+ or Si![]() O- Ca2+ bonds.
This separates the SiO2 tetrahedral from each other, which makes the mixture
more fluid and therefore more likely to form a glass after it has been melted and then
cooled. These so-called "soda-lime" glasses account for 90% of the glass
produced.
O- Ca2+ bonds.
This separates the SiO2 tetrahedral from each other, which makes the mixture
more fluid and therefore more likely to form a glass after it has been melted and then
cooled. These so-called "soda-lime" glasses account for 90% of the glass
produced.
Al2O3 is added to some glasses to increase their durability; MgO is added to slow down the rate at which the glass crystallizes. Replacing Na2O with B2O3 produces a borosilicate glass that expands less on heating. Adding PbO produces lead glasses that are ideally suited for high-quality optical glass.
The most common way of preparing a glass is to heat the mixture of sand and modifiers until it melts, and then cool it quickly so that it solidifies to produce a glass. If the cooling is rapid enough, the particles in the liquid state can't return to the original crystalline arrangement of the starting materials. Instead, they occupy randomly arranged lattice sites in which no planes of atoms can be identified. The result is an amorphous (literally: "without shape") material.
An accidental overheating of a glass furnace led to the discovery of materials known as glass-ceramics. When the glass was overheated, small crystals formed in the amorphous material that prevented cracks from propagating through the glass.
The first step toward glass-ceramics involves conventional techniques for preparing a glass. The product is then heated to 750-1150ºC, until a portion of the structure is transformed into a fine-grained crystalline material. Glass-ceramics are at least 50% crystalline after they have been heated. In some cases, the final product is more than 95% crystalline.
Because glass-ceramics are more resistant to thermal shock, cookware made of this material can be transferred directly from a hot stove burner to the refrigerator without breaking. Because they are more crystalline glass-ceramics are also slightly better at conducting heat than conventional glasses. Glass-ceramics are also stronger at high temperatures than glasses. Thus, the glass-ceramic MgO - Al2O3 - SiO2 is used to make electrical insulators that have to operate at high temperatures, such as spark plug insulators. The properties and uses of some glasses and glass-ceramics are given in the table below.
Properties and Uses of Some Glasses and Glass-Ceramics
| Composition | Property | Use | |||
| Glasses | |||||
| Al2O3, MgO, CaO, SiO2 | Translucent, chemically resistant |
Window glass, bottles | |||
| PbO, SiO2 | High refractive index | Lead crystal | |||
| B2O3, SiO2, Na2O | Acid resistant, low expansion on heating |
Pyrex | |||
| Glass Ceramics | |||||
| MgO, Al2O3, SiO2 | Insulator with high mechanical strength at high temperatures |
Spark plug insulators | |||
| CaSiO3, CaMgSi2O6, CaAl2Si2O8 |
Wear resistant | Building materials | |||
| Li2Si2O5 | Resistant to thermal shock |
Nose cones on rockets, cookware |
The term ceramic comes from the Greek word for pottery. It is used to describe a broad range of materials that include glass, enamel, concrete, cement, pottery, brick, porcelain, and chinaware. This class of materials is so broad that it is often easier to define ceramics as all solid materials except metals and their alloys that are made by the high-temperature processing of inorganic raw materials.
Ceramics can be either crystalline or glass-like. They can be either pure, single-phase materials or mixtures of two or more discrete substances. Most ceramics are polycrystalline materials, with abrupt changes in crystal orientation or composition across each grain in the structure. Ceramics can have electrical conductivities that resemble metals, such as ReO3 and CrO2.
Ceramics can also make excellent insulators, such as the glass-ceramics used in spark plugs.
One of the most distinctive features of ceramics is their resistance to being worked or shaped after they are fired. With certain exceptions, such as glass tubing or plate glass, they can't be sold by the foot or cut to fit on the job. Their size and shape must be decided on before they are fired and they must be replaced, rather than repaired, when they break.
The primary difference between ceramics and other materials is the chemical bonds that
hold these materials together. Although they can contain covalent bonds, such as the Si![]() O
O![]() Si linkages in glass, they are often characterized by ionic bonds
between positive and negative ions. When they form crystals, the strong force of
attraction between ions of opposite charge in the planes of ions make it difficult for one
plane to slip past another. Ceramics are therefore brittle. They resist compression, but
they are much weaker to stress applied in the form of bending.
Si linkages in glass, they are often characterized by ionic bonds
between positive and negative ions. When they form crystals, the strong force of
attraction between ions of opposite charge in the planes of ions make it difficult for one
plane to slip past another. Ceramics are therefore brittle. They resist compression, but
they are much weaker to stress applied in the form of bending.
The use of ceramics traces back to Neolithic times, when clay was first used to make bowls that were baked in campfires. Clay is formed by the weathering of rock to form shinglelike particles of alumina and silica that cling together when wet to form clay minerals, such as kaolinite, which has the formula Al4Si4O10(OH)8.
Today, ceramics play an important role in the search for materials that can resist thermal shock, act as abrasives, or have a better weight-strength ratio. Alumina ceramics are used for missile and rocket nose cones, silicon carbide (SiC) and molybdenum disilicide (MoSi2) are used in rocket nozzles, and ceramic tiles are used for thermal insulation to protect the Space Shuttle on re-entry through the Earth's atmosphere.
Ceramics made from uranium dioxide (UO2) are being used as the fuel elements for nuclear power plants. Ceramics are also used as laser materials, from the chromium-doped crystals that emit a coherent monochromatic pulse of light to the optics through which the light passes. BaTiO3 is used to make ceramic capacitors that have a very high capacitance. It is also used to make piezoelectric materials that develop an electric charge when subjected to a mechanical stress, which are the active elements of phonograph cartridges, sonar, and ultrasonic devices. Magnetic ceramics formed by mixing ZnO, FeO, MnO, NiO, BaO, or SrO with Fe2O3 are used in permanent magnets, computer memory, and telecommunications.
Materials Science | Metals and Unit Cells | More about Materials Science | Ceramics
Periodic Table | Glossary | Cool Applets
Gen Chem Topic Review | General Chemistry Help Homepage | Search: The general chemistry web site.