| Position of Transition Metals in the Periodic Table | Transition Metals vs. Main-Group Elements | The Electron Configuration of Transition-Metal Ions |
| Oxidation States of the Transition Metals | ||
Position of Transition Metals in the Periodic Table
The elements in the periodic table are often divided into four categories: (1) main group elements, (2) transition metals, (3) lanthanides, and (4) actinides. The main group elements include the active metals in the two columns on the extreme left of the periodic table and the metals, semimetals, and nonmetals in the six columns on the far right. The transition metals are the metallic elements that serve as a bridge, or transition, between the two sides of the table. The lanthanides and the actinides at the bottom of the table are sometimes known as the inner transition metals because they have atomic numbers that fall between the first and second elements in the last two rows of the transition metals.
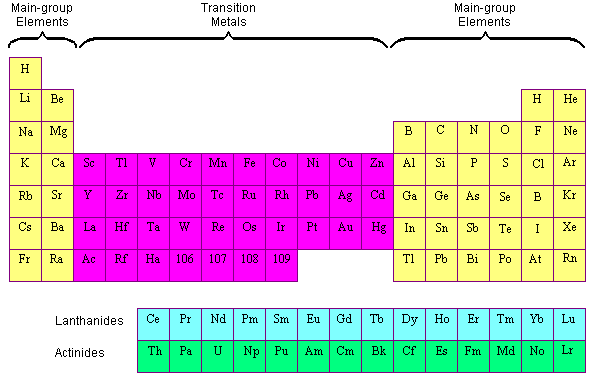
Transition Metals vs. Main-Group Elements
There is some controversy about the classification of the elements on the boundary between the main group and transition-metal elements on the right side of the table. The elements in question are zinc (Zn), cadmium (Cd), and mercury (Hg).
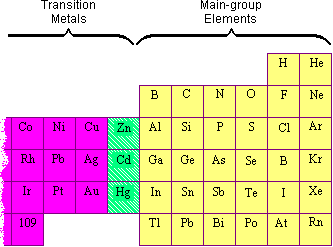
The disagreement about whether these elements should be classified as main group elements or transition metals suggests that the differences between these categories are not clear. Transition metals are like main group metals in many ways: They look like metals, they are malleable and ductile, they conduct heat and electricity, and they form positive ions. The fact the two best conductors of electricity are a transition metal (copper) and a main group metal (aluminum) shows the extent to which the physical properties of main group metals and transition metals overlap.
There are also differences between these metals. The transition metals are more electronegative than the main group metals, for example, and are therefore more likely to form covalent compounds.
Another difference between the main group metals and transition metals can be seen in the formulas of the compounds they form. The main group metals tend to form salts (such as NaCl, Mg3N2, and CaS) in which there are just enough negative ions to balance the charge on the positive ions. The transition metals form similar compounds [such as FeCl3, HgI2, or Cd(OH)2], but they are more likely than main group metals to form complexes, such as the FeCl4-, HgI42-, and Cd(OH)42- ions, that have an excess number of negative ions.
A third difference between main group and transition-metal ions is the ease with which they form stable compounds with neutral molecules, such as water or ammonia. Salts of main group metal ions dissolve in water to form aqueous solutions.
| H2O | ||||
| NaCl(s) | Na+(aq) | + | Cl-(aq) |
When we let the water evaporate, we get back the original starting material, NaCl(s). Salts of the transition-metal ions can display a very different behavior. Chromium(III) chloride, for example, is a violet compound, which dissolves in liquid ammonia to form a yellow compound with the formula CrCl3 6 NH3 that can be isolated when the ammonia is allowed to evaporate.
CrCl3(s) + 6 NH3(l)
CrCl3 6 NH3(s)
The Electron Configuration of Transition-Metal Ions
The relationship between the electron configurations of transition-metal elements and their ions is complex.
Example: Let's consider the chemistry of cobalt which forms complexes that contain either Co2+ or Co3+ ions.
The electron configuration of a neutral cobalt atom is written as follows.
Co: [Ar] 4s2 3d7
The discussion of the relative energies of the atomic orbitals suggests that the 4s orbital has a lower energy than the 3d orbitals. Thus, we might expect cobalt to lose electrons from the higher energy 3d orbitals, but this is not what is observed. The Co2+ and Co3+ ions have the following electron configurations.
Co2+: [Ar] 3d7
Co3+: [Ar] 3d6
In general, electrons are removed from the valence-shell s orbitals before they are removed from valence d orbitals when transition metals are ionized.
| Practice Problem 1: Predict the electron configuration of the Fe3+ ion. |
Because the valence electrons in transition-metal ions are concentrated in d orbitals, these ions are often described as having dn configurations. The Co3+ and Fe2+ ions, for example, are said to have a d6 configuration.
Co3+: [Ar] 3d6
Fe2+: [Ar] 3d6
Oxidation States of the Transition Metals
Most transition metals form more than one oxidation state.
Some oxidation states, however, are more common than others. The most common oxidation states of the first series of transition metals are given in the table below. Efforts to explain the apparent pattern in this table ultimately fail for a combination of reasons. Some of these oxidation states are common because they are relatively stable. Others describe compounds that are not necessarily stable but which react slowly. Still others are common only from a historic perspective.
Common Oxidation States of the First Series of Transition Metals
| Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | |||||||||||
| +1 | d10 | |||||||||||||||||||
| +2 | d3 | d5 | d6 | d7 | d8 | d9 | d10 | |||||||||||||
| +3 | d0 | d3 | d5 | d6 | ||||||||||||||||
| +4 | d0 | d3 | ||||||||||||||||||
| +5 | d0 | |||||||||||||||||||
| +6 | d0 | |||||||||||||||||||
| +7 | d0 |
One point about the oxidation states of transition metals deserves particular attention: Transition-metal ions with charges larger than +3 cannot exist in aqueous solution.
Consider the following reaction in which manganese is oxidized from the +2 to the +7 oxidation state.
| Mn2+(aq) + 4 H2O(l) |
When the manganese atom is oxidized, it becomes more electronegative. In the +7 oxidation state, this atom is electronegative enough to react with water to form a covalent oxide, MnO4-.
It is useful to have a way of distinguishing between the charge on a transition-metal ion and the oxidation state of the transition metal. By convention, symbols such as Mn2+ refer to ions that carry a +2 charge. Symbols such as Mn(VII) are used to describe compounds in which manganese is in the +7 oxidation state.
Mn(VII) is not the only example of an oxidation state powerful enough to decompose water. As soon as Mn2+ is oxidized to Mn(IV), it reacts with water to form MnO2. A similar phenomenon can be seen in the chemistry of both vanadium and chromium. Vanadium exists in aqueous solutions as the V2+ ion. But once it is oxidized to the +4 or +5 oxidation state, it reacts with water to form the VO2+ or VO2+ ion. The Cr3+ ion can be found in aqueous solution. But once this ion is oxidized to Cr(VI), it reacts with water to form the CrO42- and Cr2O72- ions.
