The Valence-Bond Approach to Bonding in Complexes
The idea that atoms form covalent bonds by sharing pairs of electrons was first proposed by G. N. Lewis in 1902. It was not until 1927, however, that Walter Heitler and Fritz London showed how the sharing of pairs of electrons holds a covalent molecule together. The Heitler-London model of covalent bonds was the basis of the valence-bond theory. The last major step in the evolution of this theory was the suggestion by Linus Pauling that atomic orbitals mix to form hybrid orbitals, such as the sp, sp2, sp3, dsp3, and d2sp3 orbitals.
It is easy to apply the valence-bond theory to some coordination complexes, such as the Co(NH3)63+ ion. We start with the electron configuration of the transition- metal ion.
Co3+: [Ar] 3d6
We then look at the valence-shell orbitals and note that the 4s and 4p orbitals are empty.
Co3+: [Ar] 3d6 4s0 4p0
Concentrating the 3d electrons in the dxy, dxz, and dyz orbitals in this subshell gives the following electron configuration.
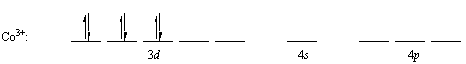
The 3dx2-y2, 3dz2, 4s, 4px, 4py and 4pz orbitals are then mixed to form a set of empty d2sp3 orbitals that point toward the corners of an octahedron. Each of these orbitals can accept a pair of nonbonding electrons from a neutral NH3 molecule to form a complex in which the cobalt atom has a filled shell of valence electrons.
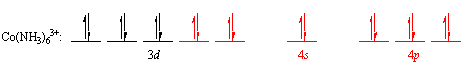
| Practice Problem 4: Use valence-bond theory to explain why Fe2+ ions form the Fe(CN)64- complex ion. |
At first glance, complexes such as the Ni(NH3)62+ ion seem hard to explain with the valence-bond theory. We start, as always, by writing the configuration of the transition-metal ion.
Ni2+: [Ar] 3d8
This configuration creates a problem, because there are eight electrons in the 3d orbitals. Even if we invest the energy necessary to pair the 3d electrons, we can't find two empty 3d orbitals to use to form a set of d2sp3 hybrids.
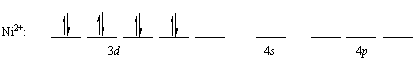
There is a way around this problem. The five 4d orbitals on nickel are empty, so we can form a set of empty sp3d2 hybrid orbitals by mixing the 4dx2-y2, 4dz2, 4s, 4px, 4py and 4pz orbitals. These hybrid orbitals then accept pairs of nonbonding electrons from six ammonia molecules to form a complex ion.
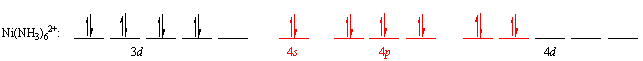
The valence-bond theory therefore formally distinguishes between "inner-shell" complexes, which use 3d, 4s and 4p orbitals to form a set of d2sp3 hybrids, and "outer-shell" complexes, which use 4s, 4p and 4d orbitals to form sp3d2 hybrid orbitals.
